The Waisman Center: Decades later, what would Harry think?
Last fall, the Waisman Center at the University of Wisconsin–Madison bid successfully for the same National Institutes of Health core grant that the late Harry Waisman first won 45 years ago.

Harry Waisman, left, with colleagues C.S. Reiquam and Nathan J. Smith, examine pathology microscope slides in a research lab.
Photo: UW-Madison Archives
The award continues the legacy of the UW–Madison pediatrician and biochemist who was a trailblazer in the scientific study and treatment of developmental disabilities.
The nearly $6 million federal award will fund the core activities of the center through its 50th year in 2016. “This was the grant Harry Waisman wrote,” notes Marsha Mailick Seltzer, a professor of social work and the center’s director since 2002. “[He] had a comprehensive vision — clinics, wet labs, testing facilities for children, education — all under the umbrella of research.”
The Waisman Center first sprang to life in 1963 as the Joseph P. Kennedy, Jr. Memorial Laboratories. In 1965, the National Institute of Child Health and Human Development selected it as one of the initial two sites in the country for the construction of a multidisciplinary center for the study of human development and what was then termed “mental retardation.”
Since the Waisman Center’s inception, science has made amazing strides. The center’s ongoing work would no doubt amaze and please Waisman, an individual whose research, among other accomplishments, helped inform our understanding of Phenylketonuria or PKU, an inherited condition that can cause severe mental retardation.
Waisman’s scientific passion and advocacy for those with developmental disorders, notes Seltzer, is the archetype for what has become one of the world’s leading centers of research, education and outreach on developmental disabilities and neurodegenerative diseases.
“Waisman was, in many respects, a visionary,” avers Seltzer. “I like to think that the Waisman Center has realized that vision, and in many ways, I believe it has. And although science has changed dramatically since Waisman’s time, how we explore and exploit new knowledge about developmental disabilities and neurodegenerative diseases, I believe, rests on the foundation he set 50 years ago.”
So, what would Harry Waisman, who died at age 58, think of the accomplishments of the center that bears his name? These 10 examples suggest that the work going on today would astonish him.
More Effective PKU Treatment
Mandatory screening for and treating the condition Phenylketonuria (PKU) was one of Waisman’s signature missions in life. Untreated, PKU leads to profound mental retardation. If the condition is identified at birth, it can be effectively treated through a special diet that helps patients mitigate the toxic effects of an amino acid that those with PKU are unable to metabolize. But the diet, explains Seltzer, leaves much to be desired; it is unappetizing and does not satisfy hunger. The diet is a lifelong exercise to keep PKU at bay, and pregnant women with PKU must conform to the diet to prevent their babies from having the condition.
Enter Waisman researchers Denise Ney, professor of nutritional science, and Sandra van Calcar, assistant professor of pediatrics, as well as Mark Etzel, a professor of food science. Etzel developed a new, more appetizing food for PKU patients, which is now in clinical trials at the Waisman Center and at the Harvard-affiliated Children’s Hospital in Boston.
“Denise’s research carries forth the legacy of Harry Waisman through prevention of disability via screening, early identification and dietary treatment,” says Seltzer. “Indeed, PKU is the only genetic disorder that can be fully treated.”
The Genetic Roots of Developmental Disability

Professor Albee Messing, left, discovered the gene that causes a rare condition in young children and later devised a test for the disease.
Photo: Jeff Miller
Since Waisman’s death in 1971, there has been a surge of discovery in genetic research, with well more than 6,000 single-gene disorders identified as the root cause of human disease, including many that cause developmental disabilities, according to Seltzer. Emblematic of such research is Waisman scientist Albee Messing’s discovery of the gene that causes Alexander disease, a rare but fatal condition that mainly affects young children. Messing, a professor of comparative biosciences in the UW–Madison School of Veterinary Medicine, has since devised a genetic test for the disease and begun teasing out the molecular mechanisms that cause symptoms and death in Alexander patients. He is currently screening existing drugs already approved for human use that may help ease the manifestations of this currently untreatable disease.
Fragile X
A condition involving changes in the X chromosome, fragile X is the most prevalent inherited developmental disability. It is caused by changes in a gene known as FMR1, where a small part of the gene’s amino-acid sequence repeats more often than normal. In most people, the segment repeats five to 40 times. Increased repetition of the sequence is associated with impaired cognitive and reproductive functions. While 200 repeats is considered a full mutation of the FMR1 gene, anything between 41 and 54 repeats, just above normal expression, falls into what researchers call the “gray zone,” where there is a borderline risk of neurological symptoms. Between 55 and 200 repeats is considered a “premutation.” Seltzer conducted the first U.S. survey of the prevalence of both the fragile X gray zone and permutation, and she found a surprisingly high prevalence of both, with the gray zone occurring in one of every 12 females and one of every 21 males, suggesting that the genetic preconditions for clinical fragile X are widespread.
Stem Cells
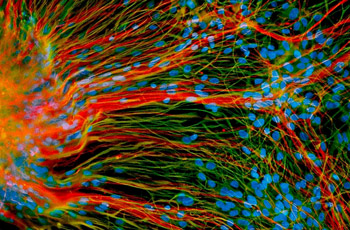
UW-Madison work with stem cells is leading to an understanding of what can go wrong at the level of a cell to cause conditions such as Down, fragile X and Rett syndromes.
Photo: Su-Chun Zhang
First tamed in 1998, embryonic stem cells — the blank slate cells that arise in early development and go on to form all 220 tissues in the human body — have since been directed in the lab dish by Su-Chun Zhang, a Waisman Center scientist and professor of neuroscience, to become some of the most fundamental building blocks of the brain. Scientists can now make neurons and astrocytes with relative ease, and they’re exploring the intricate workings of those cells and how they might be used clinically to alleviate conditions such as Parkinson’s and Lou Gehrig’s disease. What’s more, embryonic — and now induced pluripotent stem cells — are being used by Waisman scientists to create models of diseases such as Down, fragile X and Rett syndromes, as well as disorders that cause blindness and deafness. Such work promises a fundamental understanding of what goes wrong at the level of the cell to cause those conditions and may one day lead to their prevention.
The Epidemiology of Autism
Since it was first described scientifically in 1943, autism has become recognized as one of the most widespread developmental disorders in the United States and beyond. At the time the Waisman Center was founded, the first epidemiologic study of autism in the country was under way in Wisconsin by clinical professor Darold Treffert. That study found many children with autism to be institutionalized and not allowed to enroll in typical school classrooms.] It also found the prevalence of autism to be just 3.1 per 10,000 or 1 in 3,225 children.
Collaborative studies now under way at the Waisman Center find the prevalence of autism spectrum disorders in Wisconsin today to be about 7.6 per 1,000 children (1 in 131), or more than 20 times more common than it was found to be in the 1960s.
While the causes of autism and its rising prevalence are the subjects of debate, a number of notable facts and clues to the etiology of the condition have come to light through the work of Maureen Durkin, a Waisman Center researcher and professor of population health sciences. In carrying out epidemiological investigations of autism prevalence, Durkin and her colleagues identified higher socioeconomic status as a risk factor, whereas the risk of developmental disabilities overall tends to be associated with lower socioeconomic status. Other risk factors for autism identified by Durkin and her colleagues include advanced maternal and paternal ages, and earlier birth order. Much additional research is needed to translate these epidemiological studies into clinical treatments, but understanding the population trends in a necessary first step.
Seeing the Autistic Brain
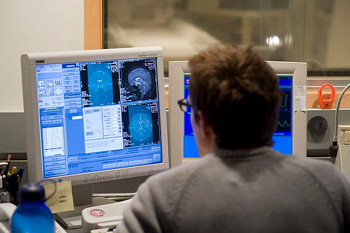
A technician reviews computer displays following an imaging test at the Waisman Center to measure a subject’s brain activity.
Photo: Jeff Miller
Although a prevalent condition, autism remains a mystery on many levels. Richard Davidson, a Waisman Center researcher and Vilas Professor of Psychology and Psychiatry, has used sophisticated brain imaging techniques to help resolve a little bit of that mystery by identifying abnormalities characteristic of autism in the amygdala, an almond-sized mass deep in the brain. Andrew Alexander, a Waisman Center researcher and an associate professor of medical physics and psychiatry, and his colleagues have shown that a sophisticated form of magnetic resonance imaging can be used to differentiate the brains of individuals with autism and those in control groups with 94 percent accuracy.
Language as Child’s Play

Professor Jenny Saffran’s research is exploring how infants develop language skills.
Photo: Jeff Miller
How infants develop language skills has been the subject of intellectual dogfights for decades. Are we hard-wired at birth to acquire language? Or is language something the brain must soak up from our environment? Psychology Professor Jenny Saffran, who directs the Waisman Center’s Infant Learning Laboratory, waded into the infant language wars as a graduate student; her work helped to show that the language skills of very young humans is — at least in part — due to sophisticated learning strategies that play out in the first year of life. Infants, Saffran found, could sort out and focus on new words embedded in strings of nonsensical language. In the Infant Learning Lab, Saffran and her team continue to divine the mysteries of how humans acquire language skills early on and how infants might become better processors of language.
Speaking Through Computers
Computers help us do many things, but they have become an essential communication lifeline for many children and others with developmental disabilities. Established at the Waisman Center in 1971 to address the needs of people who are disabled and don’t speak, the Trace Center became an innovative player in the emerging field of “augmentative communication.” With the advent of personal computers, the Trace Center helped devise, test and gain industry acceptance of many of the accessibility features that are now standard in the machines we use in our everyday lives. The Trace Center, now a part of the College of Engineering, continues to provide services through the Communication Aids and Systems Clinic at Waisman. Katie Hustad, an associate professor of communicative disorders, leads the clinic.
Growing Old with Down Syndrome
Eighty years ago, people with Down syndrome lived, on average, to age 9. Today, they live into their 50s and 60s, thanks to advances in medicine. However, people with Down syndrome tend to exhibit premature aging and dementia. Alzheimer’s, in particular, commonly begins to manifest itself by the fourth decade of life, although many do not show the changes in behavior associated with the big changes taking place in the brain.
To better forecast the possibility of dementia, Bradley Christian, a Waisman researcher and associate professor of medical physics and psychiatry, is using MRI and PET scanning techniques to identify changes in the brain that can predict which adults with Down syndrome are susceptible to dementia. Although dementia is a high risk for many with Down syndrome, Sigan Hartley, a Waisman Center researcher and assistant professor of human development and family studies, is working with Christian to document the neuropsychological changes that are associated with the changes in the brain documented by the MRI and PET scans. Together these two researchers are helping to identify pathways to healthy aging in people with Down syndrome.
Making Medicine
Taking scientific discovery to the clinic is no small task. Among the many hurdles any new therapy must clear is devising a way to manufacture agents — drugs, vaccines, vectors and cells — for clinical trials in a way that meets strict FDA standards. Waisman Biomanufacturing was set in motion under the direction of Derek Hei in 2001, and during the past decade, it has developed a portfolio of clinical products, including some of the first therapeutic human embryonic stem cells, as well as vaccines, recombinant proteins and products for gene therapy. With seven cleanroom areas capable of clinical-quality production of a suite of biological therapeutic agents, the Waisman Center has partnered widely with companies in Wisconsin, the United States and abroad, as well as with academic research centers, to provide novel agents used in medical trials.




